Function-Preserving Single Amino Acid Substitutions Shield Hematopoietic Stem and Progenitor Cells from CD117-Targeted Immunotherapy in Vivo
Romina Marone1,2, Rosalba Lepore1,2,3,4, Julia Rositzka5, Viviane Dettmer-Monaco5, Alessandro Dell’ Aglio1,2, Giuseppina Capoferri1,2, Julie Brault4, Emiel Ten Buren3, Thomas Burgold3, Anna Camus3, Christopher Divsalar4, Laura Garcia Prat3, Anna Haydn4, Felix Hermann4, Marcel Heugel3, Andreja Knezevic3, Frank Lehmann3, Valentin Do Sacramento3, Alessandro Sinopoli4, Lisa C. Wellinger4, Amélie Wiederkehr4, Saniye Yumlu3, Toni Cathomen5, Thomas Winkler3, Tatjana I. Cornu5, Stefanie Urlinger3,4, And Lukas T. Jeker1,2,3
1 Department of Biomedicine, Basel University Hospital and University of Basel, Basel, Switzerland; 2 Transplantation Immunology & Nephrology, Basel University Hospital, Basel, Switzerland; 3 Cimeio Therapeutics AG, Basel, Switzerland; 4 Ridgeline Discovery GmbH, Switzerland; 5 Institute for Transfusion Medicine & Gene Therapy, Medical Center – University of Freiburg, Freiburg, Germany
Introduction
- Current untargeted cytotoxic conditioning regimens for hematopoietic stem cell transplantation (HSCT) have been directly or indirectly associated with transplant related morbidity and mortality.
- HSPCs express the receptor tyrosine kinase c-KIT (CD117) which is activated by its ligand stem cell factor (SCF) leading to CD117 signaling that is crucial for HSPC biology.
- Blocking SCF binding prevents proliferation and leads to HSPC depletion in vivo2. Therefore, CD117 is an attractive therapeutic target as a conditioning agent prior to HSCT.
- mAb-based conditioning necessitates a washout phase of the depleting antibody before engraftment of donor HSPC and prevents post-transplant redosing.
- HSPCs engineered to be shielded from a CD117-targeted immunotherapy could overcome this limitation and enable novel HSCT approaches.
Aim
Methods
- Label-free determination of binding affinity of CD117 variants to SCF and anti-CD117 antibodies.
- In vitro mAb-mediated blocking binding and proliferation assays (TF-1, HSPC).
- Non-viral engineering of TF-1 cells and HSPCs using CRISPR-Cas9 (High fidelity Cas9 RNPs, single strand oligodeoxynucleotides (ssODN) as homology directed repair template (HDRT).
- ELISA-based p-Tyr719 signalling in TF-1 cells engineered to express CD117 variant 1.1.
- In vitro colony forming assay of HSPCs.
- In vivo: Injection of non-virally edited HSPCs into sublethally irradiated NSG mice + isotype/CIM058 injection.
Results
1. Rational design of human CD117 protein variants and their SCF and SR-1 binding
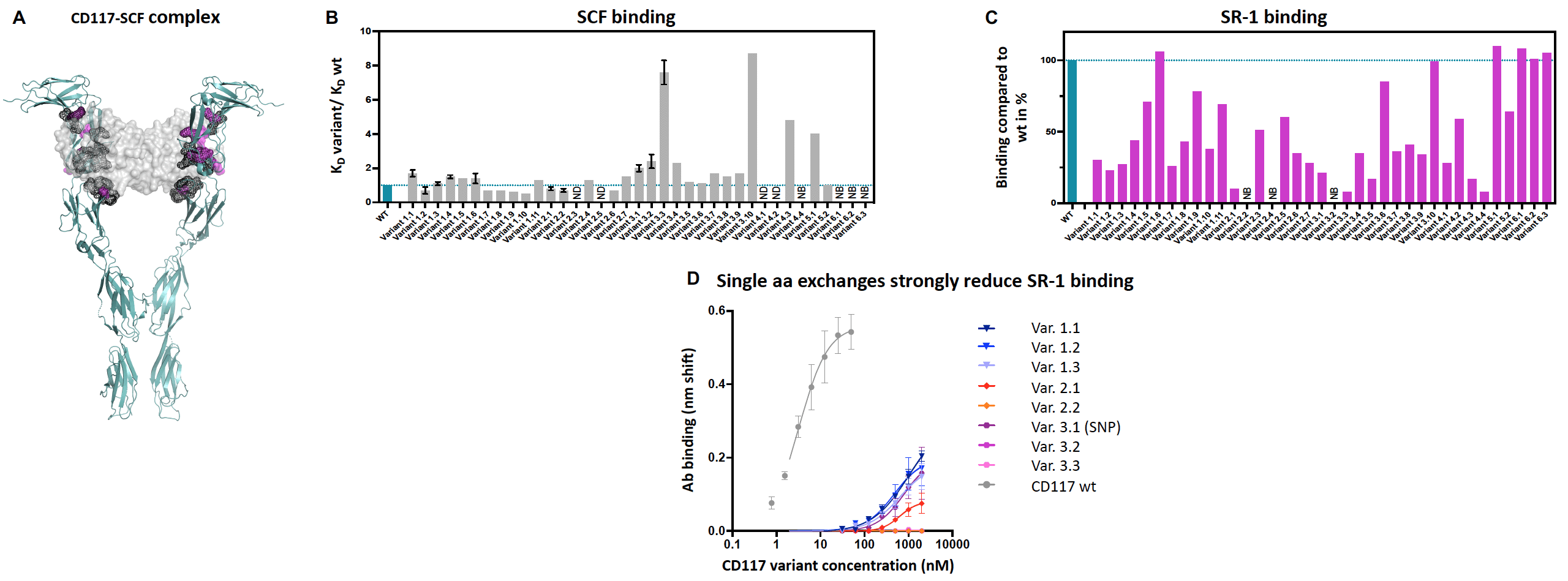
A) 3D structure of the CD117-SCF complex (PDB ID: 2e9w)1. CD117 is shown as ribbons. SCF is shown as light grey molecular surface. CD117 residues involved in SCF binding are shown as dark grey surface. CD117 residues forming the SR-1 epitope are shown as pink surface.
B) Affinity (KD) of CD117 variants binding to SCF is shown as a ratio versus WT. Values >1 (<1) indicate lower (higher) affinity to SCF. Error bars indicate the standard deviation of three measurements. NB: No Binding observed up to 1000nM of the analyte. ND: Not determined/biphasic.
C) Binding affinity of CD117 variants to SR-1 is shown as % binding vs WT. NB: No binding observed up to 500nM of the analyte.
D) Dose titration of select CD117 variants to test residual SR-1 binding at very high analyte concentrations. CD117 variants were chosen based on SR-1 binding reduction and preserved SCF binding.
2. Generation of highly potent SCF-blocking antibodies
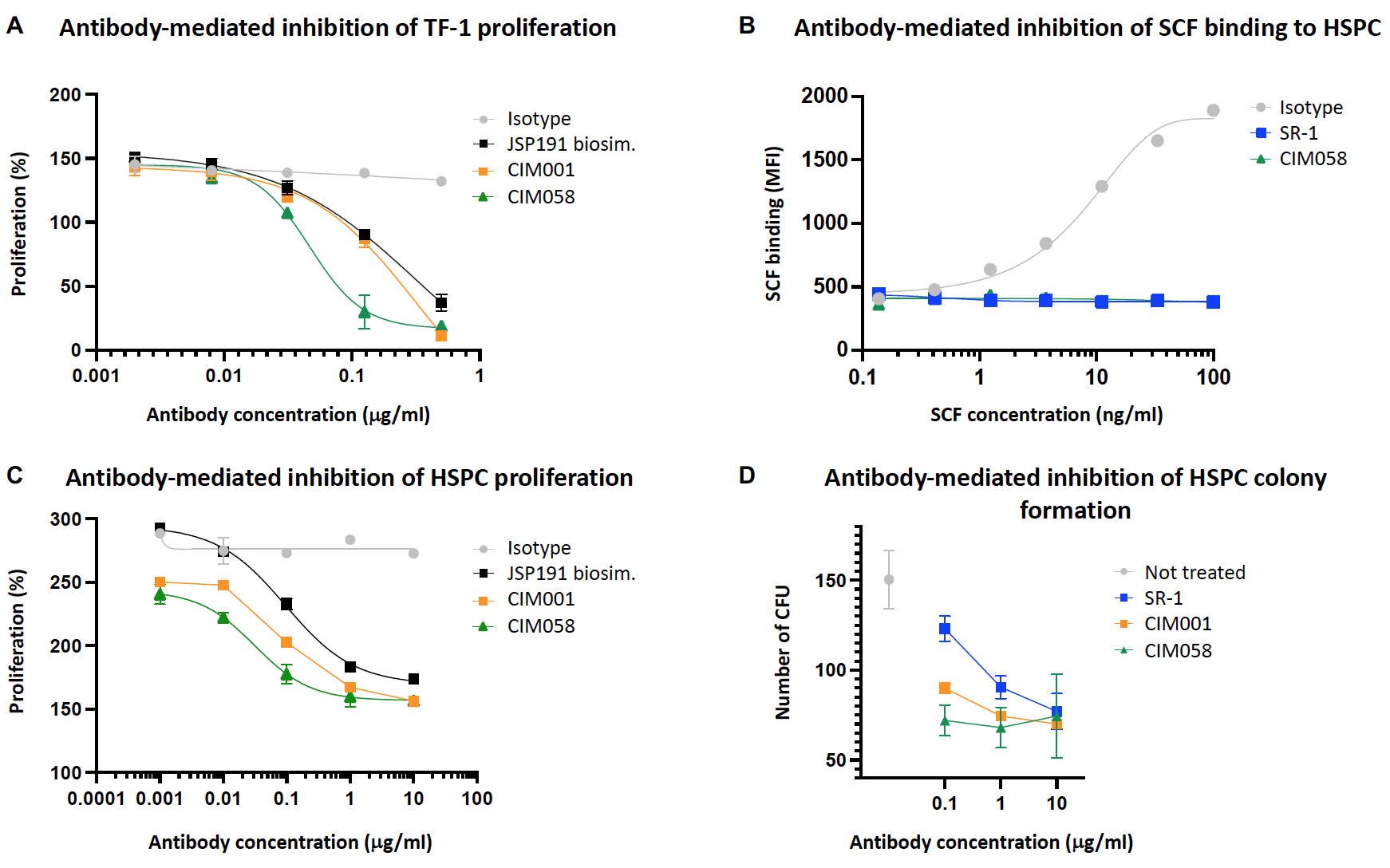
Antibodies used:
- Isotype control;
- SR-1 (known SCF mouse blocking mAb);
- JSP191 biosimilar (JSP191: humanized SR-1);
- CIM001 (proprietary humanized SCF blocking mAb);
- CIM058 (proprietary humanized and engineered SCF blocking mAb).
A) Incubation of the SCF-dependent cell line TF-1 with anti-CD117 antibodies results in dose-dependent inhibition of proliferation.
B) Incubation of HSPCs with anti-CD117 antibodies blocks SCF binding.
C) Incubation of mobilized CD34+ HSPCs with anti-CD117 antibodies results in reduced proliferation.
D) HSPCs were plated on methylcellulose and allowed to grow for 2 weeks in the presence of anti-CD117 antibodies. Colonies were counted. Anti-CD117 antibodies dose-dependently reduced colony formation.
3. Engineered TF-1: normal SCF binding, proliferation and c-Kit phosphorylation
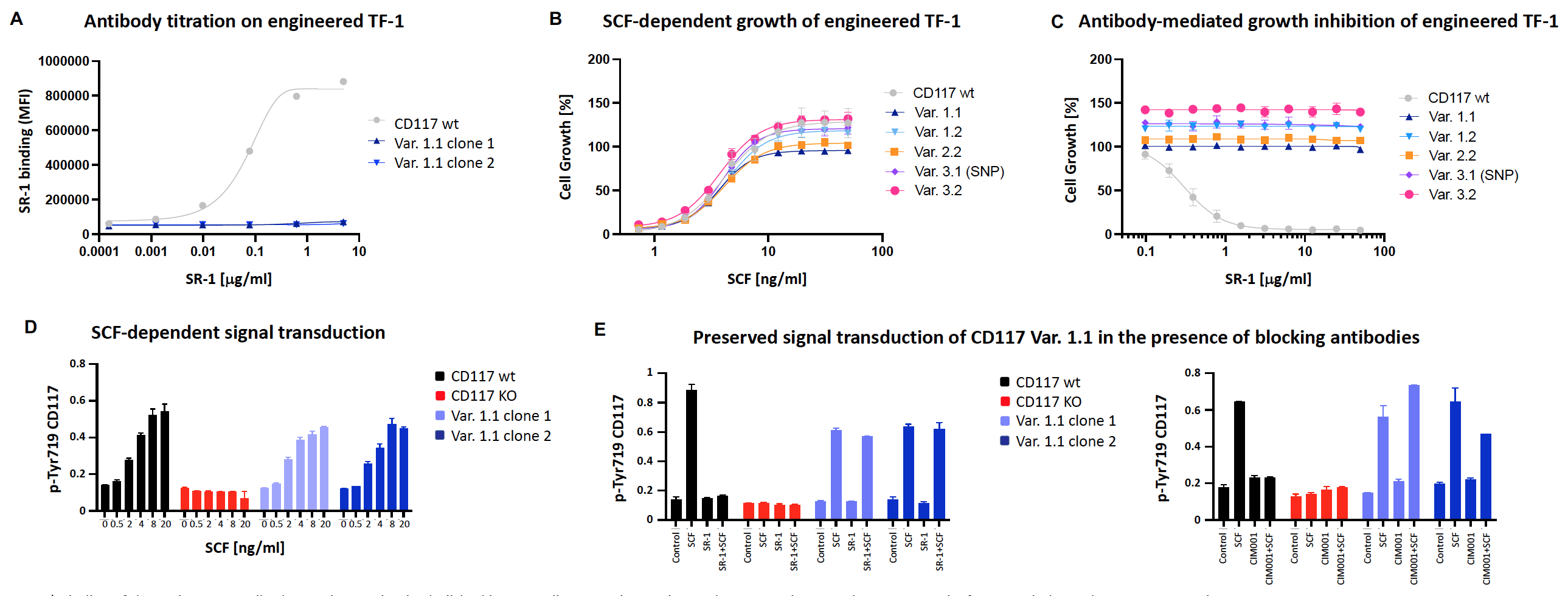
A) Binding of the anti-CD117 antibody SR-1 is completely abolished in TF-1 cells expressing engineered CD117 variant1.1. Shown are results from two independent TF-1 Var1.1 clones.
B) SCF dose-dependent proliferation of TF-1 cells expressing engineered CD117 variants is comparable (EC50) to cells with wildtype receptor.
C) Anti-CD117 antibody (SR-1) blocks SCF-mediated growth of TF-1 cells expressing wildtype but not engineered variants.
D) CD117 phosphorylation at position Tyr719 upon increasing concentrations of SCF (ELISA).
E) CD117 phosphorylation at position Tyr719 upon anti-CD117 antibody treatment is not blocked in TF-1 cells expressing the engineered variant 1.1; SCF: 0.1μg/ml; antibody: 0.5μg/ml (SR-1 or CIM001).
4. In vivo CIM058-mediated HSPC depletion and engraftment + shielding of HSPC Var.1.1
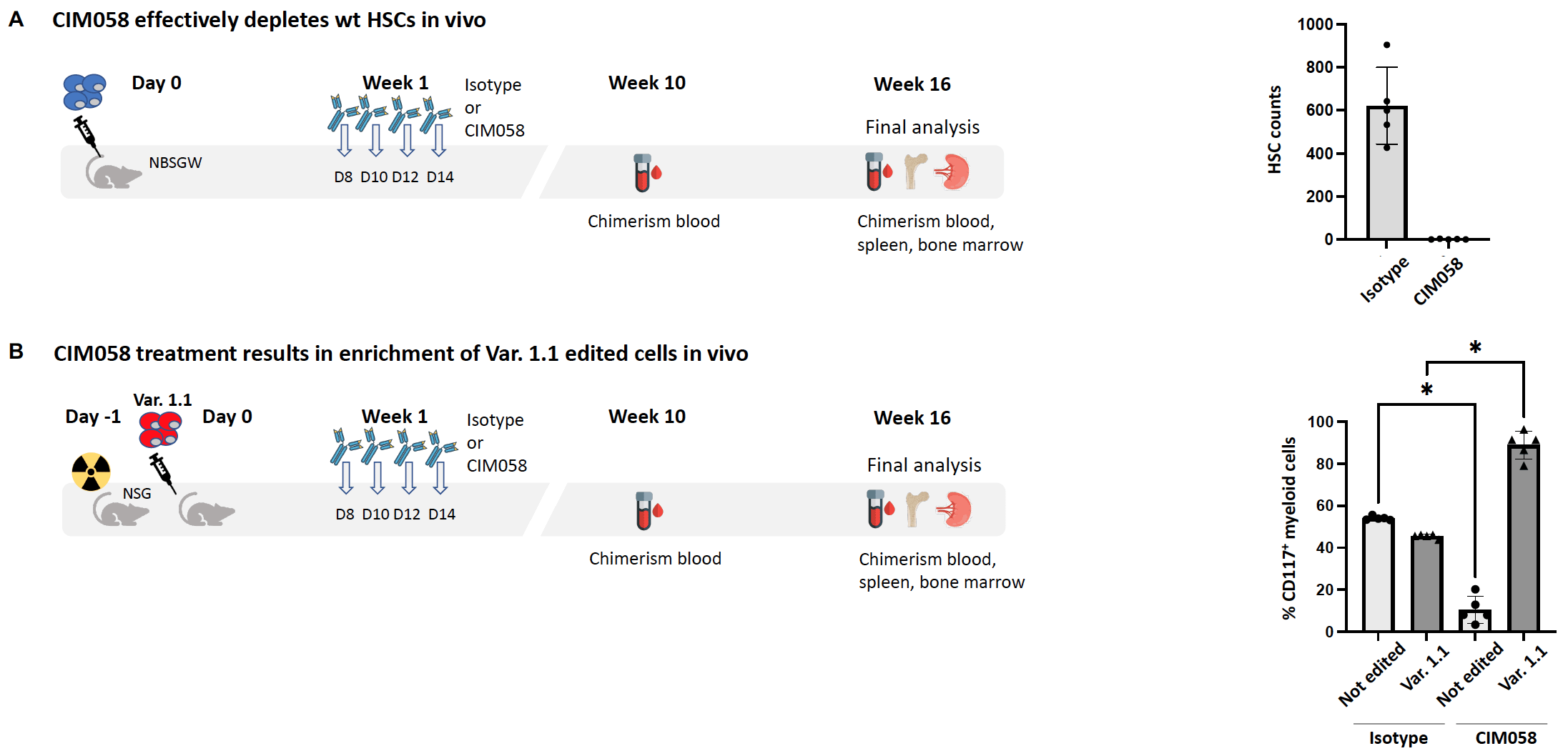
B) NSG mice irradiated one day before injection with 1 Mio gene-edited HSPCs. One week after cell injection the mice received
4x CIM058 i.v.. 16 weeks later the mice were euthanized and blood, spleen and bone marrow were analysed by flow cytometry. Results from bone marrow shown.
Conclusions
- Identified CD117 variants with normal SCF binding but reduced SR-1 binding.
- Generation of potent anti-CD117 SCF-blocking antibodies which inhibit TF-1 + HSPC proliferation and colony formation.
- TF-1 cells expressing the CD117 variants display SCF-dependent growth comparable to wt cells. Moreover, SCF-dependent signaling is intact in the presence of the SCF-blocking antibodies while KO cells and wt cells are blocked.
- Edited HSPCs engraft, differentiate in vivo but are shielded from mAbs.
References
- Yuzawa S et al. Structural Basis for Activation of the Receptor Tyrosine Kinase KIT by Stem Cell Factor. Cell 2007; 130: 323-334
- Pang WW et al. Anti-CD117 antibody depletes normal and myelodysplastic syndrome human hematopoietic stem cells in xenografted mice. Blood 2019; 133: 2069-2078
Acknowledgements
- Research Core Facilities (animal husbandry, flow cytometry) at the University of Basel and Department of Biomedicine.
- Funding from European Research Council (ERC) European Union’s Horizon 2020 research and innovation programme (grant agreement No. 818806)
