Base-Edited HSPCs Are Shielded from Targeted CD33 Therapy but Preserve CD33 Expression
1Department of Biomedicine, University of Basel, Basel, Switzerland 2Transplantation Immunology & Nephrology, Basel University Hospital, Basel, Switzerland 3Cimeio Therapeutics AG, Basel, Switzerland
Introduction
- CD33 is expressed in >90% of patients with acute myeloid leukemia (AML)
- Due to higher expression on leukemic blasts compared to their healthy counterparts, CD33 is an attractive target and already used in clinical routine (antibody drug
conjugate gemtuzumab ozogamicin) - Additionally, there are clinical trials evaluating allogeneic hematopoietic cell transplantation (HCT) with CD33 knock-out cells to avoid hematotoxicity. However,
longterm outcome of a CD33 deficient hematopoiesis remains unclear - Our group and others recently demonstrated that single amino acid changes can protect hematopoietic stem and progenitor cells (HSPCs) from targeted therapies while maintaining their function1,2,3
- The adenine base editor ABE8e allows for targeted introduction of A > G changes
Aim
Identification and characterization of a base-editable CD33 variant, that maintains function while loosing binding to a therapeutic CD33 antibody. Thereby, we create a tumor-specific antigen allowing targeted therapy for AML after allogeneic HCT without depletion of HSPCs.

Methods
- Alanine scan epitope mapping to identify single amino acid substitutions within the extracellular domain of CD33 maintaining protein structure
- Affinity screening by bio-layer interferometry (BLI) and biophysical characterization of variants by assessing melting temperature and monomer content
- Base editing screen using ABE8e_SpRY and 21 tiled sgRNAs
- Readout after editing by assessing binding to therapeutic CD33 antibody and CD33 control antibody via flow cytometry and Sanger sequencing of bulk cells
- NGS of sorted cell populations after base editing
- In vitro differentiation and colony forming assay of baseedited HSPCs
Results
1. Biophysical characterization of CD33 protein variants harboring single amino acid substitutions

A: Schematic domains of CD33, containing one C2 and one V domain B: 3D Structure of CD33 extracellular domain (PDB ID: 5IHB). Residues involved in the binding to antibody 1 are shown in sphere based on a full ECD alanine scan C: Binding affinity of CD33 variants to antibody 1 as % binding versus WT measured by BLI. Blue circles show no binding observed up to 500nM D: Monomeric content in % of purified CD33 extracellular domain E: Melting temperature of purified CD33 extracellular domain histidine tagged proteins.
2. Base editing of human CD34+ HSPCs in vitro
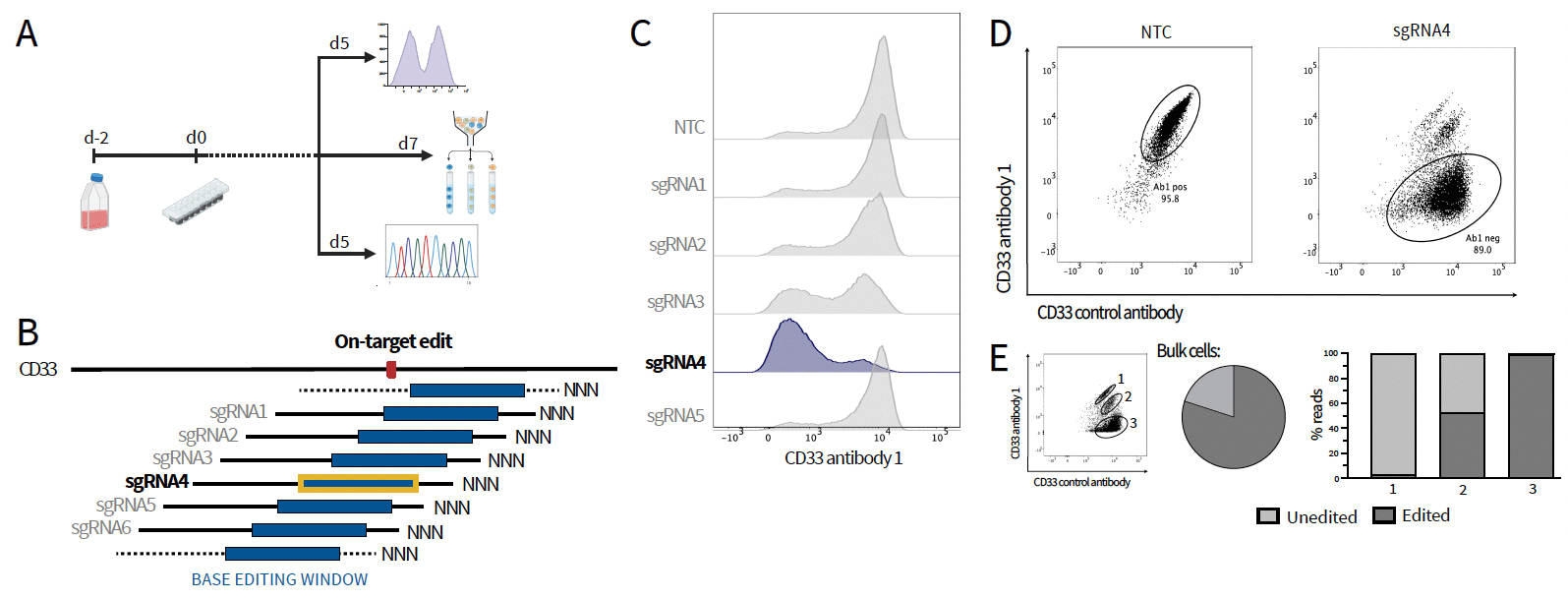
A: Timeline of base editing in HSPCs B: Base editing screen by tiling of 21 sgRNAs around the intended edit C: Histograms of CD33 target epitope antibody binding after editing of human CD34+ HSPCs with different sgRNAs in comparison to non-target control (NTC) D: Flow cytometry of edited human CD34+ HSPCs, staining with therapeutic and control CD33 antibody E: Sequencing of sgRNA4 edited bulk (Sanger sequencing) and sorted (NGS) cells, % edited and unedited cells.
3. Differentiation of sgRNA4 edited HSPCs in vitro

A: Colony-forming unit assay (CFU) with NTC and sgRNA4 edited human CD34+ HSPCs, image after 14 days B: Quantification of colony forming assays (two technical replicates) C: Sanger Sequencing of DNA extracted from single colonies from sgRNA4 edited cells in CFU, assignment to hetero- or homocygously edited cells by % of edited cells using EditR D: In vitro differentiation assay with NTC and sgRNA4 edited human HSPCs, readout with FACS after 14 days E: Quantification of three technical replicates of in vitro differentiation assay comparing myeloid and erythroid differentiation of cells.
Conclusions
- We identified a base-editable CD33 variant showing loss of antibody 1 binding while maintaining binding to a control CD33 antibody and unaltered biophysical properties compared to wildtype CD33
- HSPCs expressing this variant show a differentiation potential comparable to NTC edited HSPCs in CFU assay and in vitro differentiation assay
- Studies to further characterize the differentiation of edited cells in vivo as well as tumor models to investigate selective killing of tumor cells while preserving edited HSPCs are ongoing
References
- Marone R, Landmann E, Devaux A, et al. Epitope-engineered human hematopoietic stem cells are shielded from CD123-targeted immunotherapy. J Exp Med. 2023;220(12):e20231235. doi:10.1084/jem.20231235
- Wellhausen N et al. Epitope base editing CD45 in hematopoietic cells enables universal blood cancer immune therapy. Sci Transl Med. 2023;15(714):eadi1145. doi:10.1126/scitranslmed.adi1145
- Casirati G et al. Epitope editing enables targeted immunotherapy of acute myeloid leukaemia. Nature. 2023;621(7978):404-414. doi:10.1038/s41586-023-06496-5
Contact Information
Department of Biomedicine, Basel University Hospital and University of Basel, Basel, Switzerland lukas.jeker@unibas.ch
Acknowledgements
- Flow cytometry facility at the University of Basel and Department of Biomedicine
- Funding from European Research Council (ERC) European Union’s Horizon 2020 research and innovation programme (grant agreement No. 818806 (LTJ))
- Sponsored Research Collaboration Agreement (University of Basel/Cimeio Therapeutics AG (LTJ))
